Comparison of manual sperm analysis (WHO 5th) with two automated systems (DOWNLOAD HERE)
Lammers J, Mansour W, Jean M, Barriere P, Freour T
ART laboratory, University Hospital of Nantes, France
Introduction
Semen analysis is the first diagnostic tool to evaluate the male factor in an infertile couple. Conventional manual semen analysis is the routine method in most Assisted Reproductive Technology laboratories, but this method suffers from subjectivity and lack of standardization. Even though the recently published WHO 5th edition manual recommendations (2010) should improve the analytical performance of manual semen analysis, existing high performance automated analysis systems can provide an accurate technical alternative.
Objective
The aim of this study was to compare conventional manual sperm analysis performed according to WHO 5th edition manual (2010), Computer-Assisted Sperm Analysis (CASA, CEROS Sperm Analyzer, Hamilton Thorne) and a fully automated sperm analyzer based on electro-optical and computer algorithm technology (SQA-V Sperm Quality Analyzer, Medical Electronic Systems) in terms of standardization, accuracy and precision.
Materials and methods
All infertile men who received routine fertility evaluations at the andrology laboratory between February and April 2011 were prospectively included in the trial based on the following criteria: semen sample volume >2.5 ml (in order to allow assessments by all three methods).
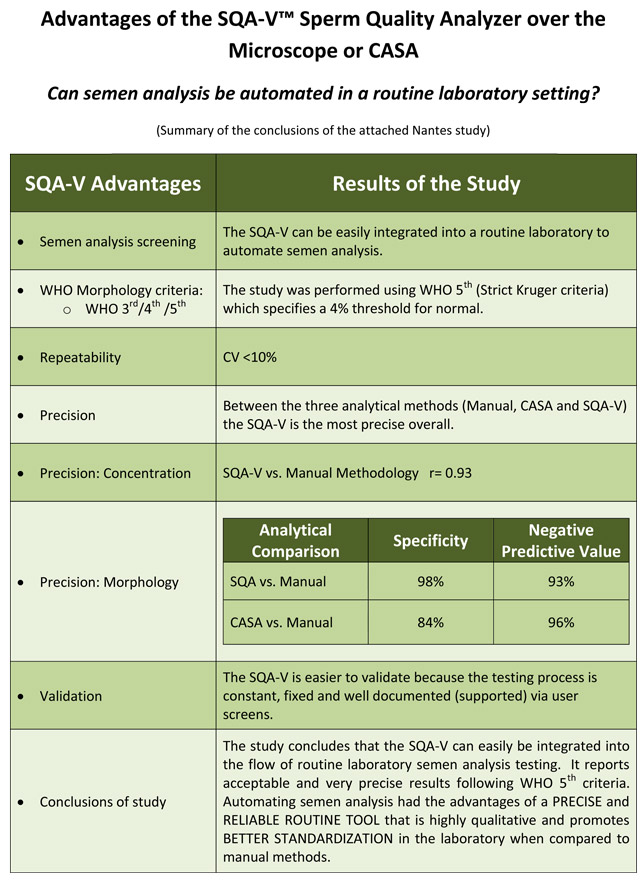
Sperm concentration, total sperm number, motility, progressive motility, motile sperm concentration (MSC), progressively motile sperm concentration (PMSC) and normal morphology (WHO 5th Kruger strict criteria, 4% threshold) were first assessed simultaneously and independently by two operators. Secondly sperm analysis was performed in duplicates by both automated systems (see workflow below).
Statistical evaluation was then performed (MedCalc software) to analyze mean values, coefficients of variation, correlation, mountain plot distribution and ROC curve parameters: specificity (Sp) and negative predictive values (NPV) for all three methods.
Results
A total of 250 patient samples were included in the study. 246 of 250 samples were compared for Sperm Concentration, Total Sperm Number and MSC. Four samples were excluded due to a testing problem. 224 of 250 samples were compared for other semen parameters. 26 samples were excluded from comparison for these parameters because of methodological difficulties due to azoospermia or severe oligospermia. Correlation coefficients of both automated systems with manual analysis were very high for sperm concentration, total sperm number, MSC, and PMSC (r>=0.93 for all parameters). The best precision was obtained with the SQA-V for all semen variables (CV<10%). All three methods showed good agreement, except for CASA MSC and PMSC that were systematically higher than reported by the other two methods. Distribution of automated systems differences versus manual assessment was analyzed by mountain plots. The SQA-V and CASA plots were comparable for concentration, but CASA plots for motility-related parameters tended to be slightly wider, compared with SQA-V. Concerning morphology, Sp and NPV of SQA-V and CASA versus manual assessment were: SpSQA – 98%, NPVSQA – 93% and SpCASA – 84%, NPVCASA – 96% respectively.
Conclusions
Both CASA and SQA-V were easy to integrate into the laboratory routine and demonstrated an acceptable agreement versus manual semen analysis performed according to WHO 5th ed. manual. The best precision among all three methods was shown by the SQA-V system. Both automated systems, and particularly SQA-V, gave accurate morphology results. Automated sperm analysis systems can be considered accurate tools for routine sperm analysis, providing high quality results and allowing better standardization than manual analysis.



